Abstract
Background: Flow and volume metrics of pulmonary function have been found to vary with ascent to high altitude and acute mountain sickness (AMS). This study aimed to corroborate prior findings and determine whether the pathophysiological changes associated with acute AMS were reflected in pulmonary function tests (PFT).
Methods: The study was conducted as part of a randomized controlled trial comparing budesonide to acetazolamide to placebo for the prevention of AMS in August 2016. Healthy adults were enrolled at 1,240 m (4,100 ft), drove to 3,545 m (11,700 ft), then hiked to and slept at 3,810 m (12,500 ft). PFT were measured using standard technique with a portable pneumotachometer at low altitude and again at sleeping altitude the evening of, and morning after ascent. The device was calibrated before each round of measurements.
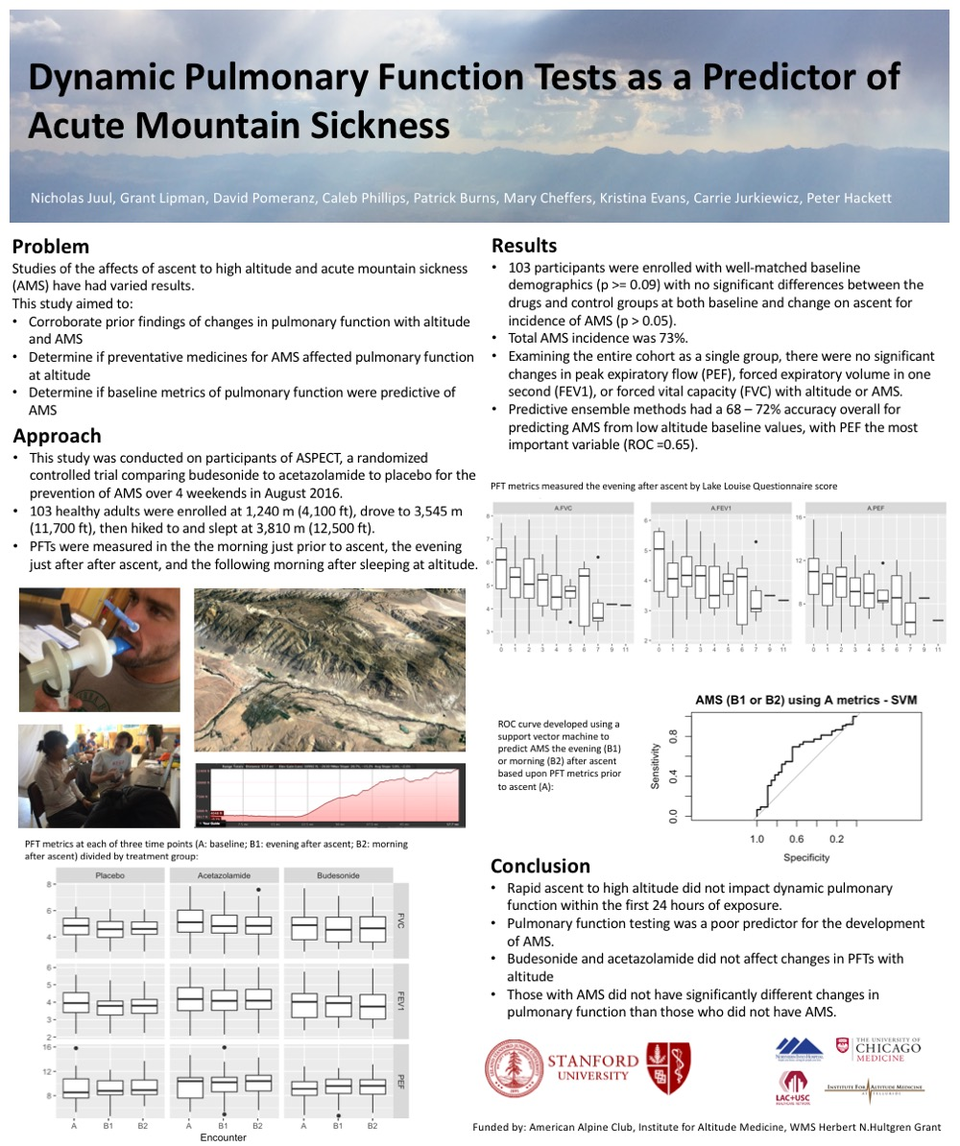
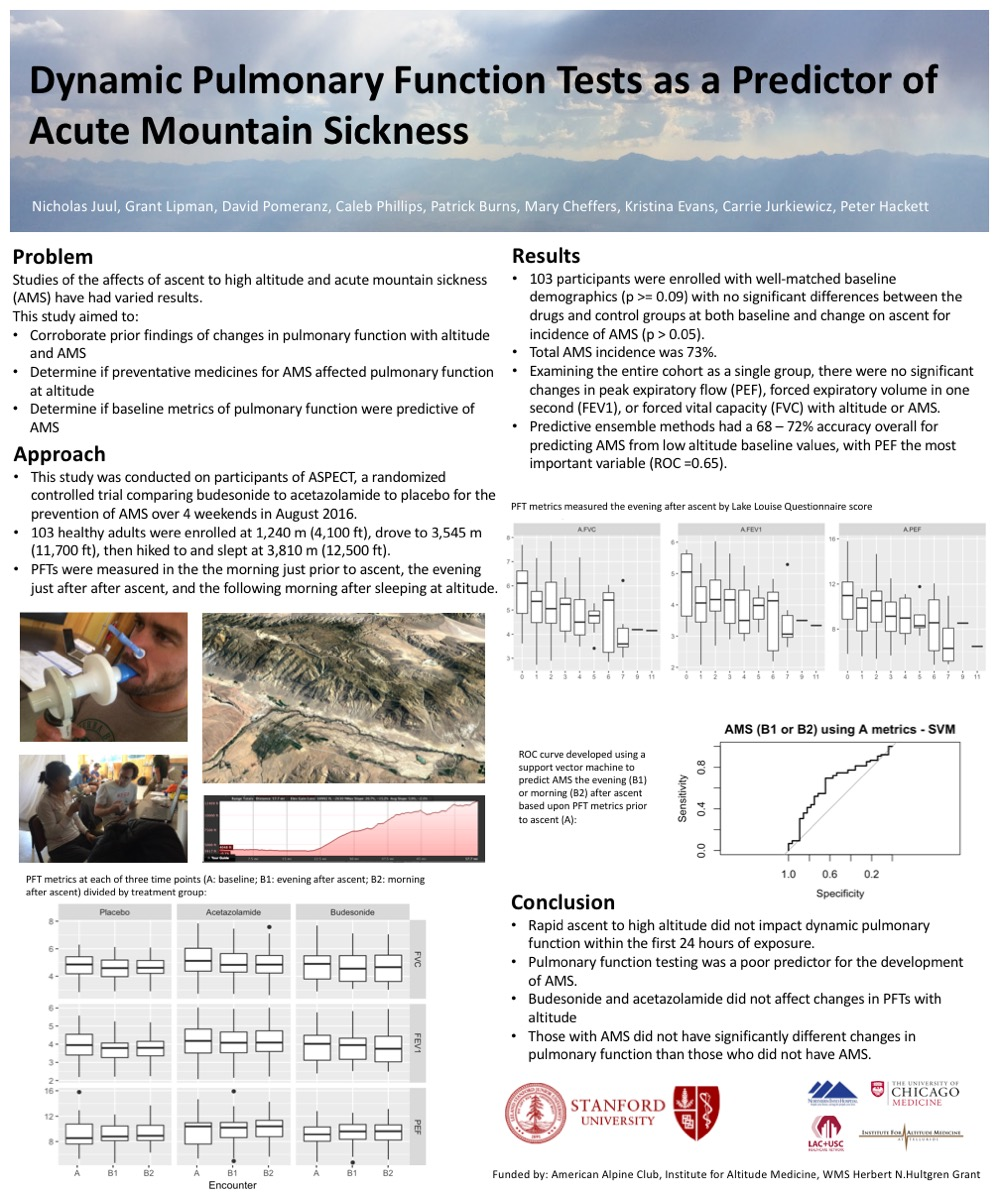
Results: Results: 103 participants were enrolled with well-matched baseline demographics (p > 0.09) with no significant differences between the drugs and control groups at both baseline and change on ascent for incidence of AMS (p > 0.05). AMS prevalence was 73%. Examining the entire cohort as a single group, there were no significant changes in peak expiratory flow (PEF), forced expiratory volume in one second (FEV1), or forced vital capacity (FVC) on ascent or in those diagnosed with AMS. Analyzed in combination, PFT had a 72% accuracy for predicting AMS from low altitude baseline, with PEF the most important variable (ROC = 0.65).
Conclusion: Rapid ascent to high altitude did not impact dynamic pulmonary function. Pulmonary function testing overall had a weak prediction for the development of AMS, with peak expiratory flow the most important variable with good accuracy.
Funding sources: American Alpine Club, Institute for Altitude Medicine, Wilderness Medical Society Herbert N. Hultgren Grant