Abstract
Background
The apicoplast is found in Plasmodium and other Apicomplexan parasites, and is unique as it has originated from secondary endosymbiosis, making it evolutionarily distinct from host cell organelles. Since it is unique to parasites and essential for survival, it is a high-value drug target as inhibitors can selectively kill the parasite without harming the host.
Protein kinases and phosphatases play a crucial role in the regulation of the metabolic processes in the apicoplast as they regulate protein activity through reversible phosphorylation. The kinases catalyze the addition of phosphate groups to serine, threonine, or tyrosine residues, while phosphatases remove these modifications. This process governs enzyme activity, protein localization, protein–protein interactions, and signal transduction pathways that are essential for metabolism, cell cycle progression, and organelle signaling. Consequently, apicoplast-localized kinases represent promising regulatory nodes and potential therapeutic targets in Plasmodium falciparum, as kinases are easy to block with medicine and are historically highly druggable.
Methods
The FIKK9.1 protein sequence was obtained from PlasmoDB and UniProt, and analyzed using ProtParam to determine physicochemical properties. Prosite, CDD, CDART, SMART, and the Superfamily were utilized to identify and confirm the conserved domains and signature motifs. The transcriptomic data from PlasmoDB were examined to assess stage-specific expression across the parasite life cycle. Structural characterization was performed through homology modeling using SWISS-MODEL and compared with AlphaFold predictions. Potential small-molecule inhibitors were identified from ChEMBL-NTD and evaluated through molecular docking using SWISS-Dock.
Results
ProtParam analysis predicted that FIKK9.1 is a stable, soluble protein with physicochemical properties consistent with enzymatic activity. Domain analysis identified a conserved serine/threonine protein kinase catalytic domain spanning residues approximately 172–539, including ATP-binding residues and a catalytically active-site motif, which support its classification as a kinase. SMART and BLAST analyses demonstrated strong conservation of FIKK9.1 across Plasmodium species and its lack of close human homologs. Structural modeling revealed a kinase fold suitable for inhibitor binding, and the docking studies identified candidate kinase inhibitors with favorable predicted binding orientations within the active site.
Conclusion
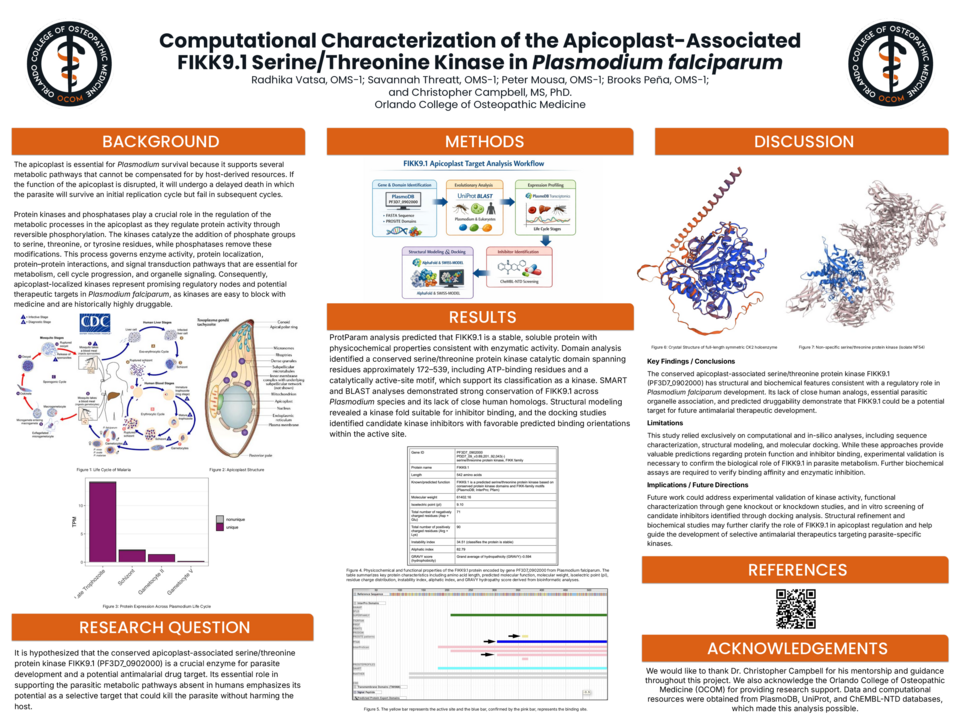
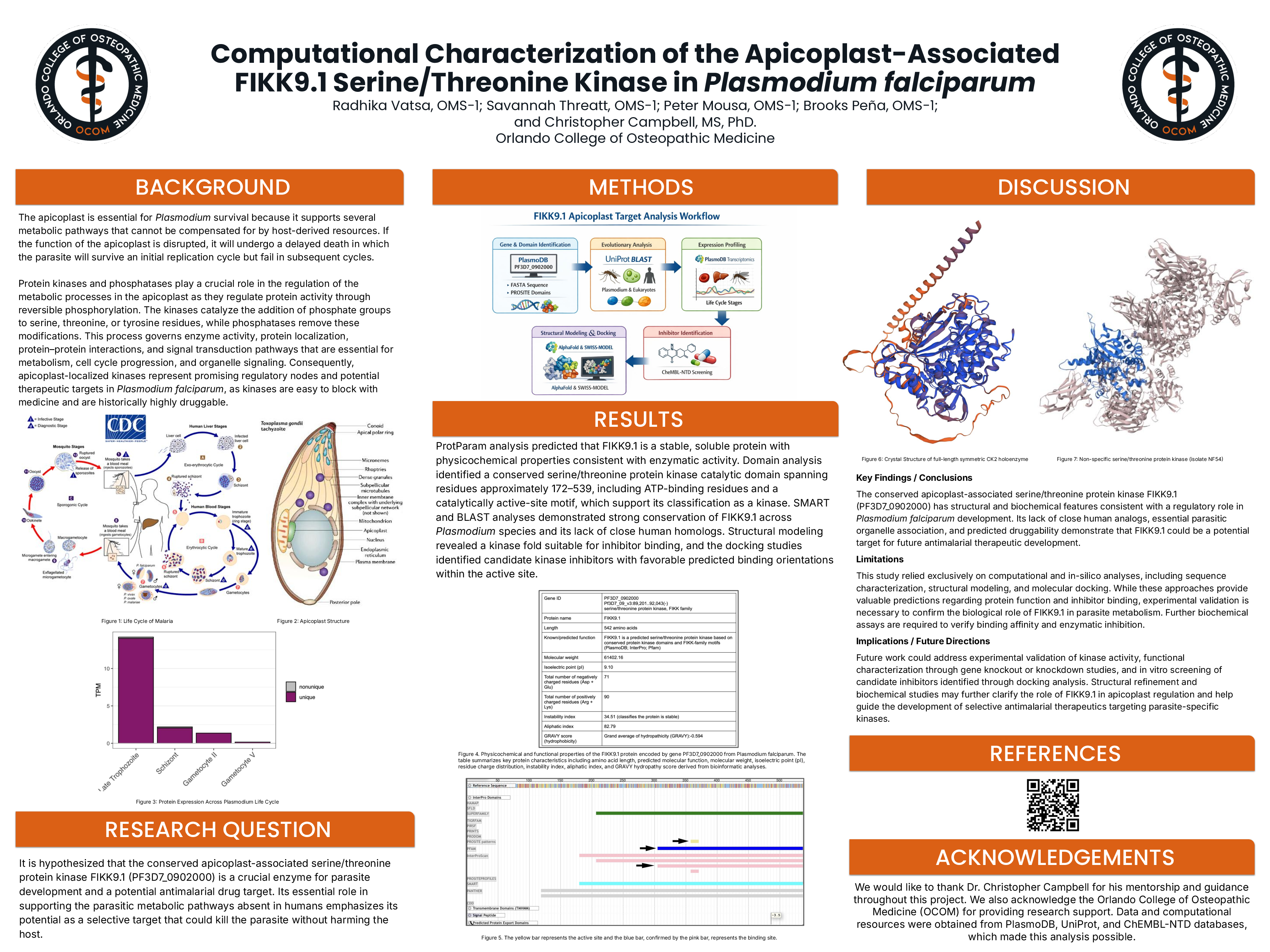
The conserved apicoplast-associated serine/threonine protein kinase FIKK9.1 (PF3D7_0902000) has structural and biochemical features consistent with a regulatory role in Plasmodium falciparum development. Its lack of close human analogs, essential parasitic organelle association, and predicted druggability demonstrate that FIKK9.1 could be a potential target for future antimalarial therapeutic development.