Abstract
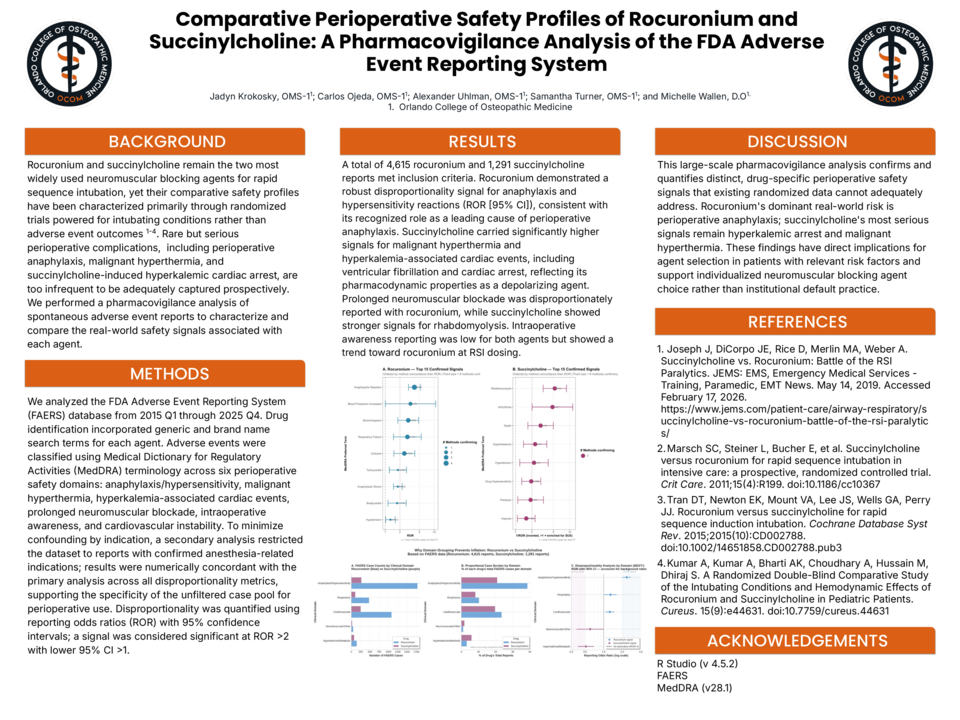
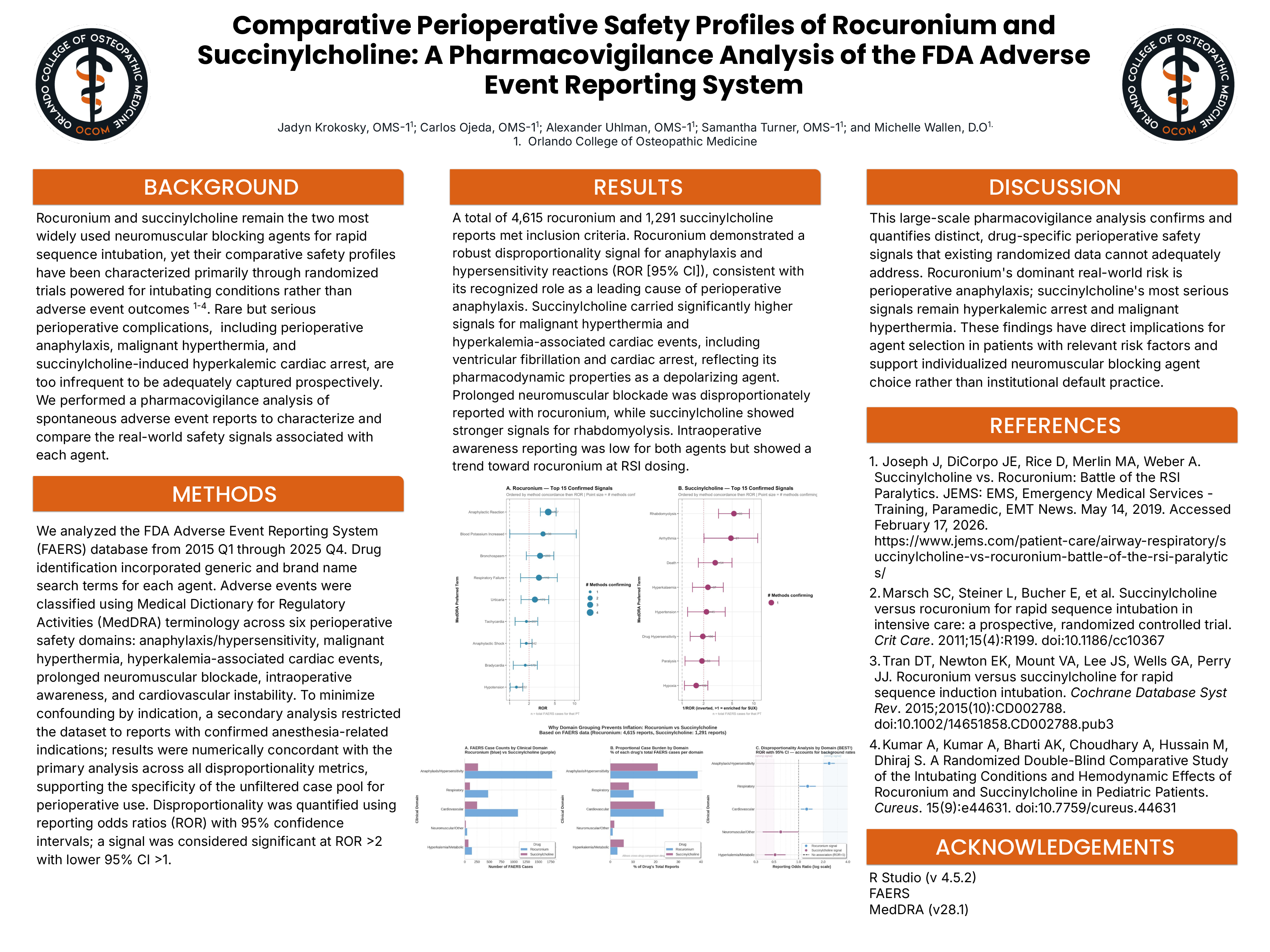
BACKGROUND: Rocuronium and succinylcholine remain the two most widely used neuromuscular blocking agents for rapid sequence intubation, yet their comparative safety profiles have been characterized primarily through randomized trials powered for intubating conditions rather than adverse event outcomes. Rare but serious perioperative complications, including perioperative anaphylaxis and hyperkalemic cardiac arrest, are too infrequent to be adequately captured prospectively. We performed a pharmacovigilance analysis of spontaneous adverse event reports to characterize and compare the real-world safety signals associated with each agent. METHODS: We analyzed the FDA Adverse Event Reporting System (FAERS) database from 2015 Q1 through 2025 Q4. Drug identification incorporated generic and brand name search terms for each agent. Adverse events were classified using Medical Dictionary for Regulatory Activities (MedDRA) terminology across six perioperative safety domains: anaphylaxis/hypersensitivity, malignant hyperthermia, hyperkalemia-associated cardiac events, prolonged neuromuscular blockade, intraoperative awareness, and cardiovascular instability. To minimize confounding by indication, a secondary analysis restricted the dataset to reports with confirmed anesthesia-related indications; results were numerically concordant with the primary analysis across all disproportionality metrics, supporting the specificity of the unfiltered case pool for perioperative use. Disproportionality was quantified using reporting odds ratios (ROR) with 95% confidence intervals; a signal was considered significant at ROR >2 with lower 95% CI >1. RESULTS: A total of 4,615 rocuronium and 1,291 succinylcholine reports met inclusion criteria. Rocuronium demonstrated a robust disproportionality signal for anaphylaxis and hypersensitivity reactions (ROR [95% CI]), consistent with its recognized role as a leading cause of perioperative anaphylaxis. Succinylcholine carried significantly higher signals for malignant hyperthermia and hyperkalemia-associated cardiac events reflecting its pharmacodynamic properties as a depolarizing agent. Prolonged neuromuscular blockade was disproportionately reported with rocuronium, while succinylcholine showed stronger signals for rhabdomyolysis. Intraoperative awareness reporting was low for both agents but showed a trend toward rocuronium at RSI dosing. DISCUSSION: This large-scale pharmacovigilance analysis confirms and quantifies distinct, drug-specific perioperative safety signals that existing randomized data cannot adequately address. Rocuronium's dominant real-world risk is perioperative anaphylaxis; succinylcholine's most serious signals remain hyperkalemic arrest and malignant hyperthermia. These findings have direct implications for agent selection in patients with relevant risk factors and support individualized neuromuscular blocking agent choice rather than institutional default practice.