Abstract
Background: Tezepelumab (Tezspire®) and dupilumab (Dupixent®) are widely used biologics for moderate-to-severe asthma, targeting anti-TSLP and anti-IL-4Rα pathways, respectively.¹⁻² Patients with severe asthma carry elevated baseline cardiovascular risk, yet real world cardiac safety profiles remain incompletely characterized.³ Despite FDA approvals in 2021 and 2018, no direct post marketing pharmacovigilance comparison of cardiac adverse events has been conducted. FAERS provides a large scale spontaneous reporting database suited for hypothesis-generating safety signal detection.⁴
Methods: FAERS was queried for all asthma-indication adverse event reports for both drugs. Disproportionality analyses used ROR with 95% CIs, PRR, and BCPNN/IC. A positive signal required ROR lower 95% CI > 1, PRR ≥ 2, and IC025 > 0.⁵⁻⁶ All other FAERS drugs served as the background comparator, with signals interpreted relative to each drug's reporting volume.
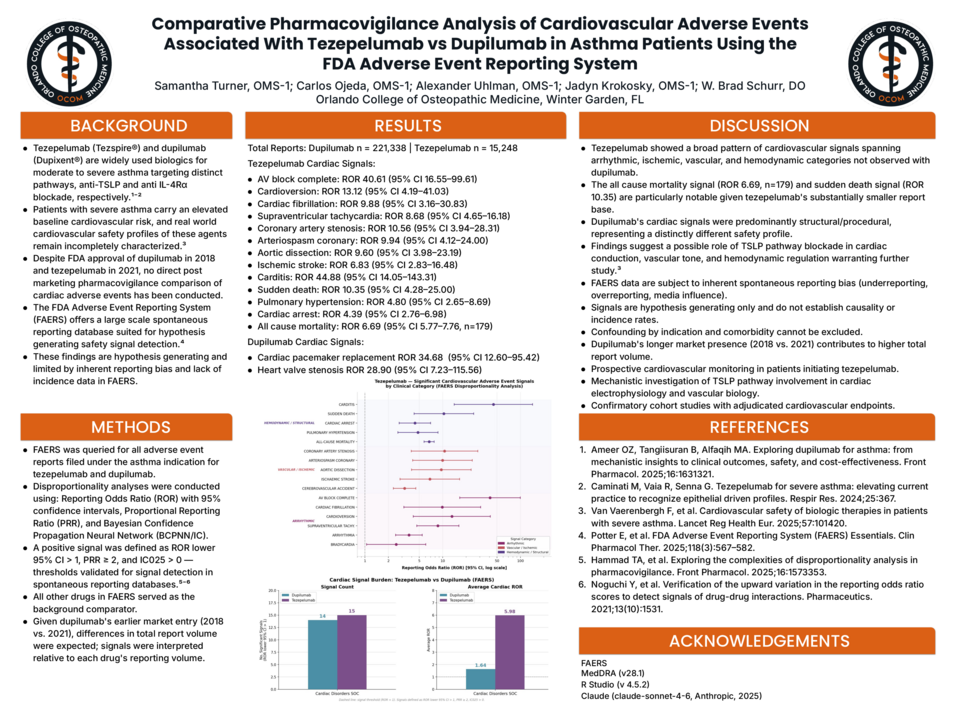
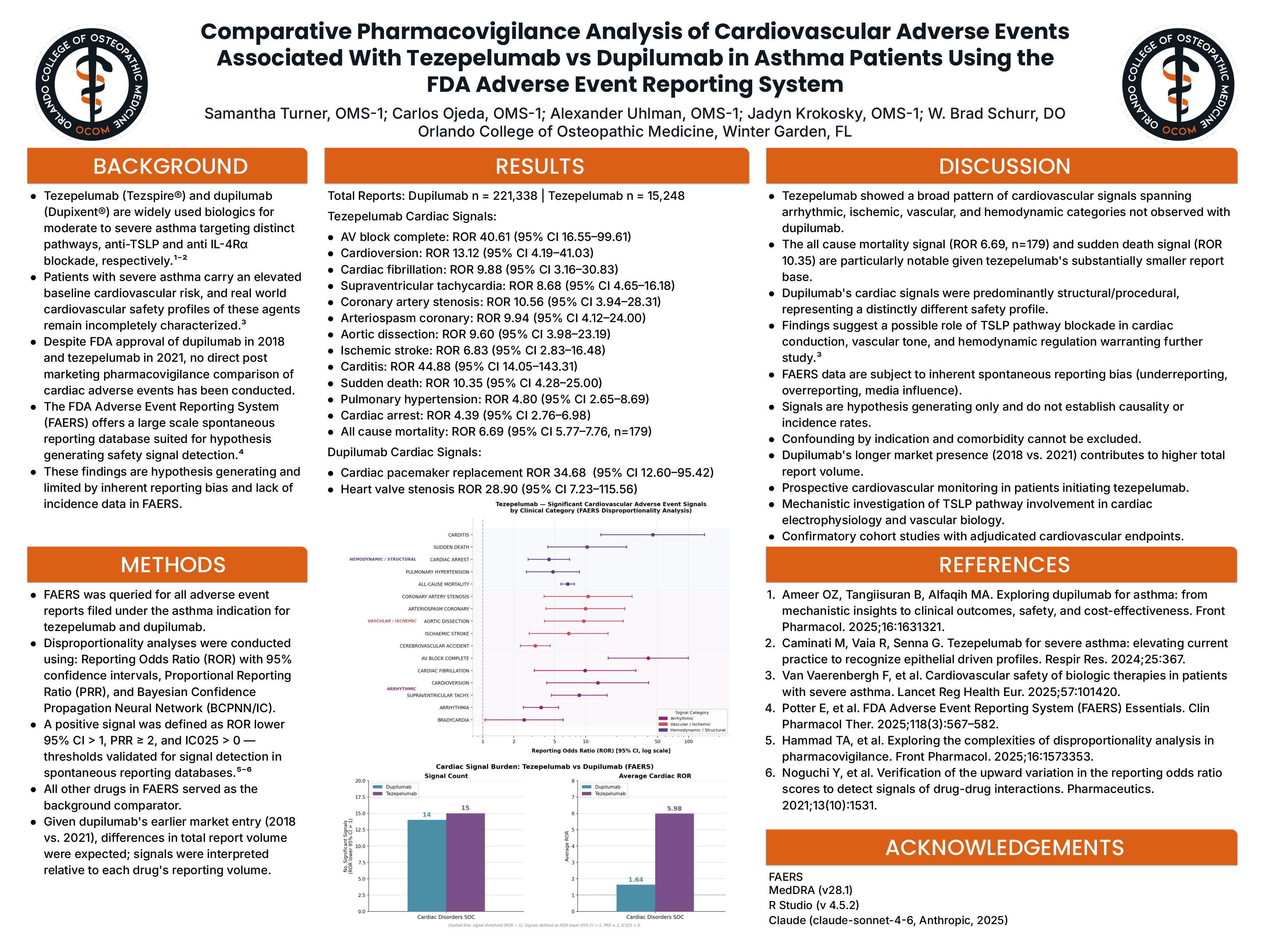
Results: Dupilumab had 221,338 reports; tezepelumab had 15,248. Despite ~14× fewer reports, tezepelumab showed a substantially higher cardiovascular signal burden across three domains:
- Arrhythmic: AV block complete (ROR 40.61, 95% CI 16.55–99.61), cardioversion (13.12, 4.19–41.03), cardiac fibrillation (9.88, 3.16–30.83), SVT (8.68, 4.65–16.18), arrhythmia (3.68, 2.48–5.45), bradycardia (2.52, 1.05–6.06)
- Vascular/Ischemic: Coronary artery stenosis (10.56, 3.94–28.31), coronary arteriospasm (9.94, 4.12–24.00), aortic dissection (9.60, 3.98–23.19), ischemic stroke (6.83, 2.83–16.48), cerebrovascular accident (3.24, 2.32–4.52)
- Hemodynamic/Structural: Carditis (44.88, 14.05–143.31), sudden death (10.35, 4.28–25.00), pulmonary hypertension (4.80, 2.65–8.69), cardiac arrest (4.39, 2.76–6.98), all-cause mortality (6.69, 5.77–7.76, n=179)
- Dupilumab signals were predominantly structural cardiac pacemaker replacement (ROR 34.68, 95% CI 12.60–95.42) and heart valve stenosis (28.90, 7.23–115.56) with no significant signals for arrest, arrhythmia, vascular events, or mortality.
Conclusion: This FAERS analysis reveals a broad cardiovascular adverse event signal pattern associated with tezepelumab not observed with dupilumab. Spanning arrhythmic, ischemic, vascular, and hemodynamic categories, including sudden death and all-cause mortality, these findings support prospective cardiovascular monitoring in tezepelumab recipients and warrant further investigation into TSLP pathway blockade.³-5 Findings are hypothesis generating and limited by FAERS reporting bias and absence of incidence data.