Abstract
Background: The underlying mechanisms associated with the onset and progression of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS), a condition characterized by symptoms such as debilitating fatigue or easy fatigability, memory problems, muscle and joint pain, gastrointestinal issues, neurological problems, and hormonal imbalance remains unclear.
Objectives: The main objectives of this research proposal are to evaluate 1) sex differences in circulating immune cells’ gene expression in ME/CFS patients, 2) sex differences in ME/CFS patients in response to stress modeled by exercise challenge, and 3) the need to consider sex differences in diagnosis and treatment of ME/CFS.
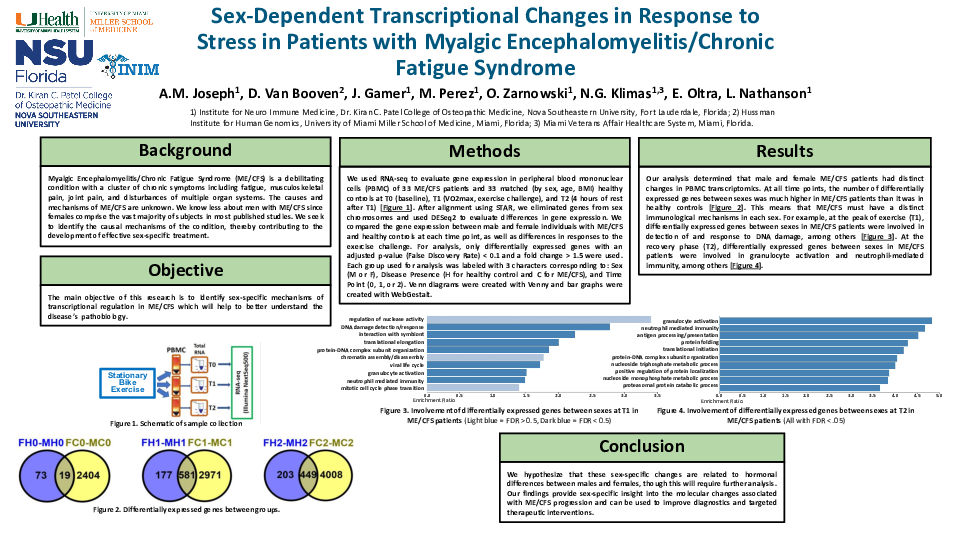
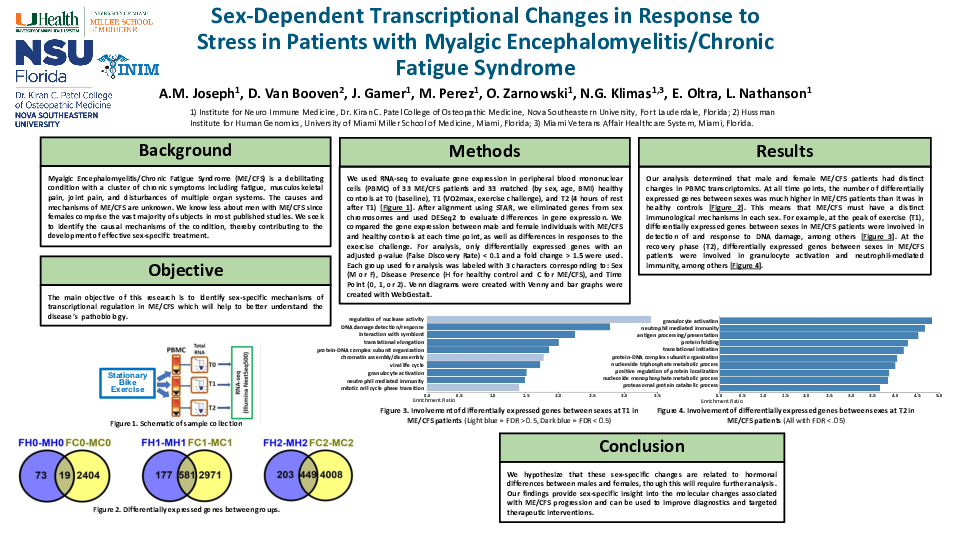
Methods: We evaluated differential gene (DG) expression (fold change > 1.5 in either direction, FDR < 0.1) by RNA-seq in circulating lymphocytes of 20 ME/CFS female patients, 14 ME/CFS male patients and 40 sex-, age- and BMI-matched healthy controls (HC) at T0 (baseline), T1 (exercise, maximal exertion) and T2 (4 hours of recovery after T1). After removing genes from sex chromosomes, analysis was done separately in men and women between ME/CFS patients and HC at each time point and in response to exercise (between time points for each group).
Results: At baseline we found 20 DG between ME/CFS patients and HC in females and 160 DG in males. The largest difference in response to exercise in men and women with ME/CFS was between T2 and T1 time points. At these time points 395 DG were the same in men and women, 167 DG were only in men, and 174 only in women. Functional analysis revealed that at baseline in males with ME/CFS the most affected were regulation of leukocyte activation (-log10(P)=6.99) and regulation of cell adhesion (-log10(P)=5.26) compared to HC, while regulation of viral process (-log10(P)=5.57) and cellular response to hypoxia (-log10(P)=4.12) were the most affected in females with ME/CFS compared to HC.
Conclusion: Identification of sex-specific biomarkers and therapeutic targets of ME/CFS can provide insight into sex-specific disease onset and progression, which will lead to optimal therapeutic intervention for such challenging disease.