Abstract
Objective: Our objective was to assess the methodologic quality of the descriptions of randomization methods used to allocate participants to comparison groups in randomized controlled trials.
Introduction: The randomization process is considered among the most important components of a randomized control trial (RCTs) and a core advantage of RCTs.
Method: We conducted a cross-sectional review of phase 3 clinical trials reported in Clinicaltrials.gov. Beginning at all records available (n = 345,278), we included studies only listed for stage 3 RCTs in the U.S. National Library of Medicine database. A total of 1528 protocols were identified as of June 1, 2020. Exclusion criteria involved no protocol listed or non-randomized studies, of which 517 were excluded and 693 text articles excluded due to unclear methods of randomization. Inclusion criteria involved randomization methods based on "A review of randomization methods in clinical trials" by Berger and Antsygina.1 Each study protocol was extracted to identify the randomization methods described by three independent reviewers.
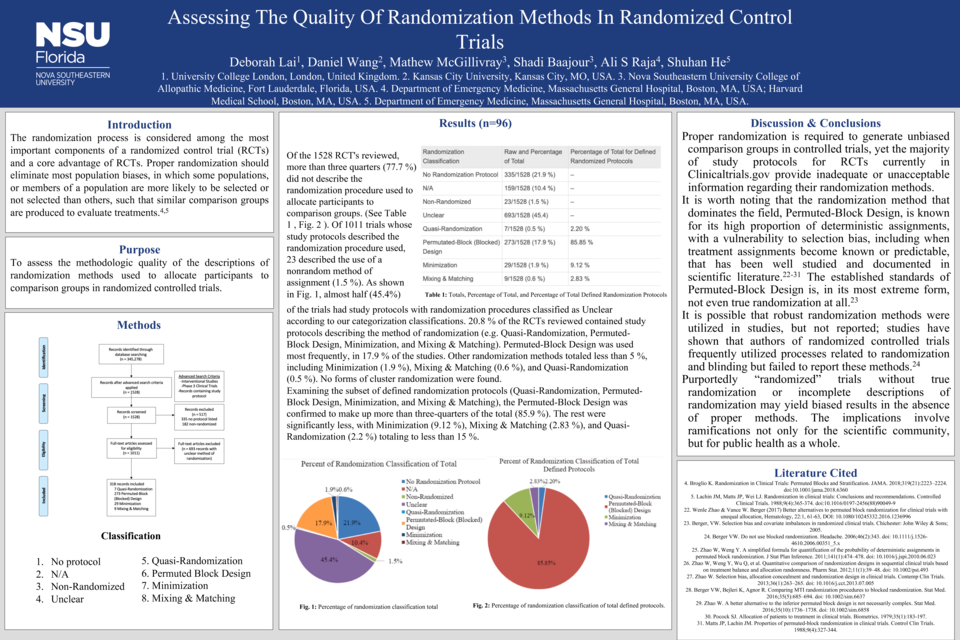
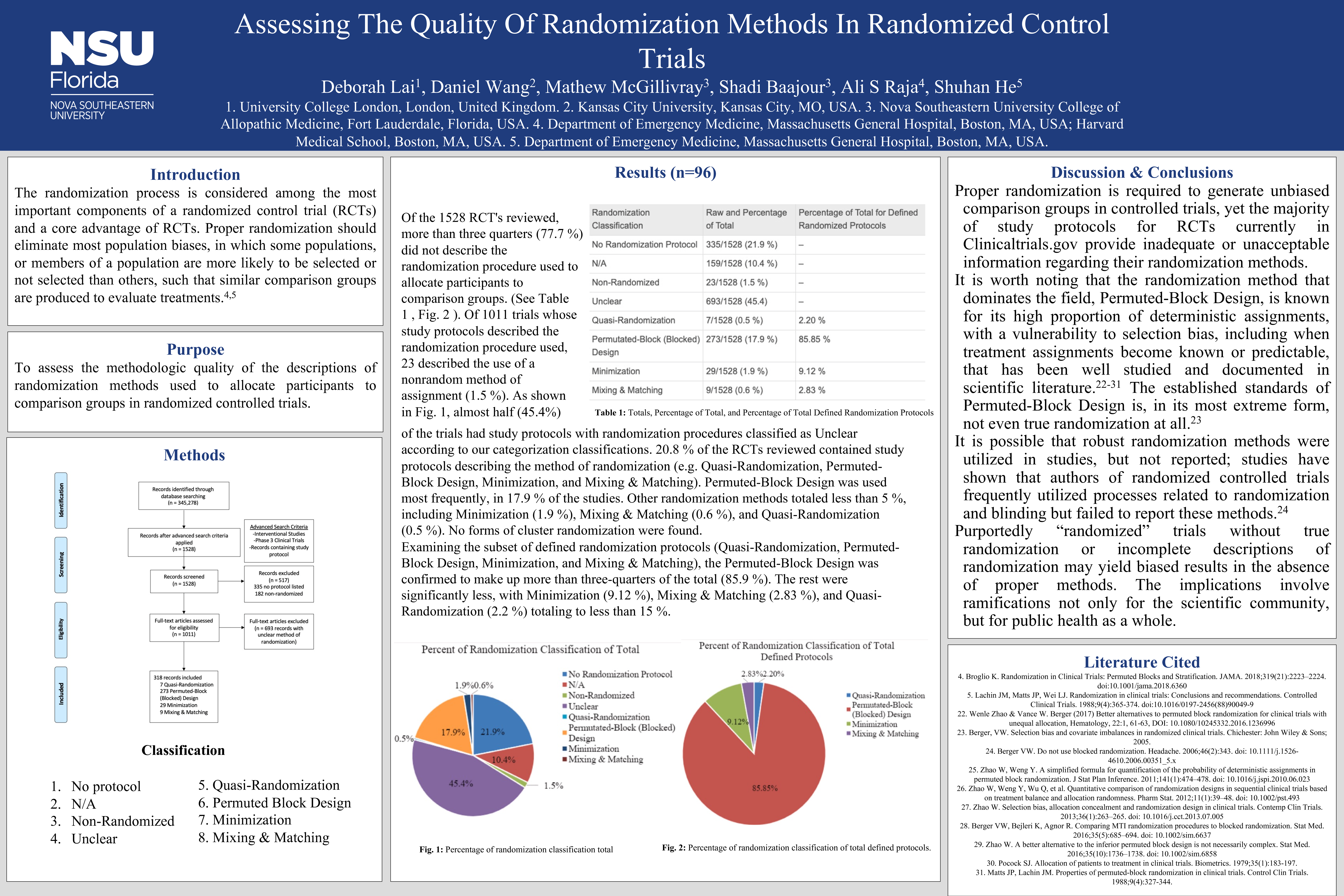
Outcomes: Only 20.8 % of the study protocols described a method for randomly assigning participants to groups. Of this subset that defined protocols, the Permuted-Block Design was used most often (85.9 %). More than three-quarters of all study protocols (77.7 %) provided incomplete descriptions about the type of randomization method.
Discussion: Proper randomization is required to generate unbiased comparison groups in controlled trials, yet the majority of study protocols for RCTs currently in Clinicaltrials.gov provide inadequate or unacceptable information regarding their randomization methods.