Abstract
Introduction
Amidst constantly evolving information on SARS-CoV-2 infection and clinical outcomes is a need to identify and protect vulnerable populations like infected obstetric patients and their neonates. Patients and physicians require further data to make informed decisions on their care and management.
Purpose
The objective of this study was to examine the studies available on the clinical outcomes of neonates born to mothers infected with SARS-CoV-2 and apply descriptive statistics to the data.
Method
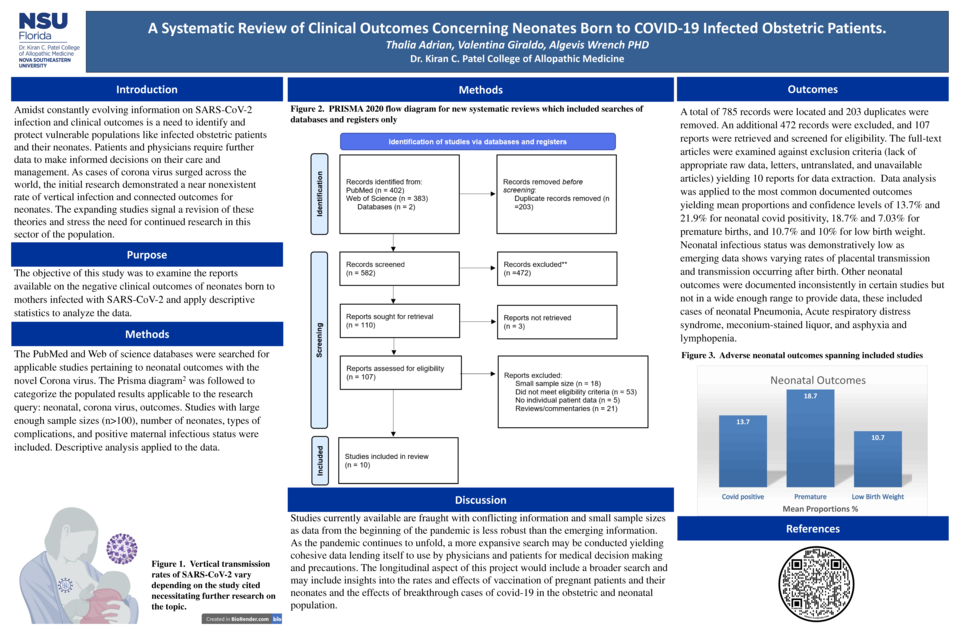
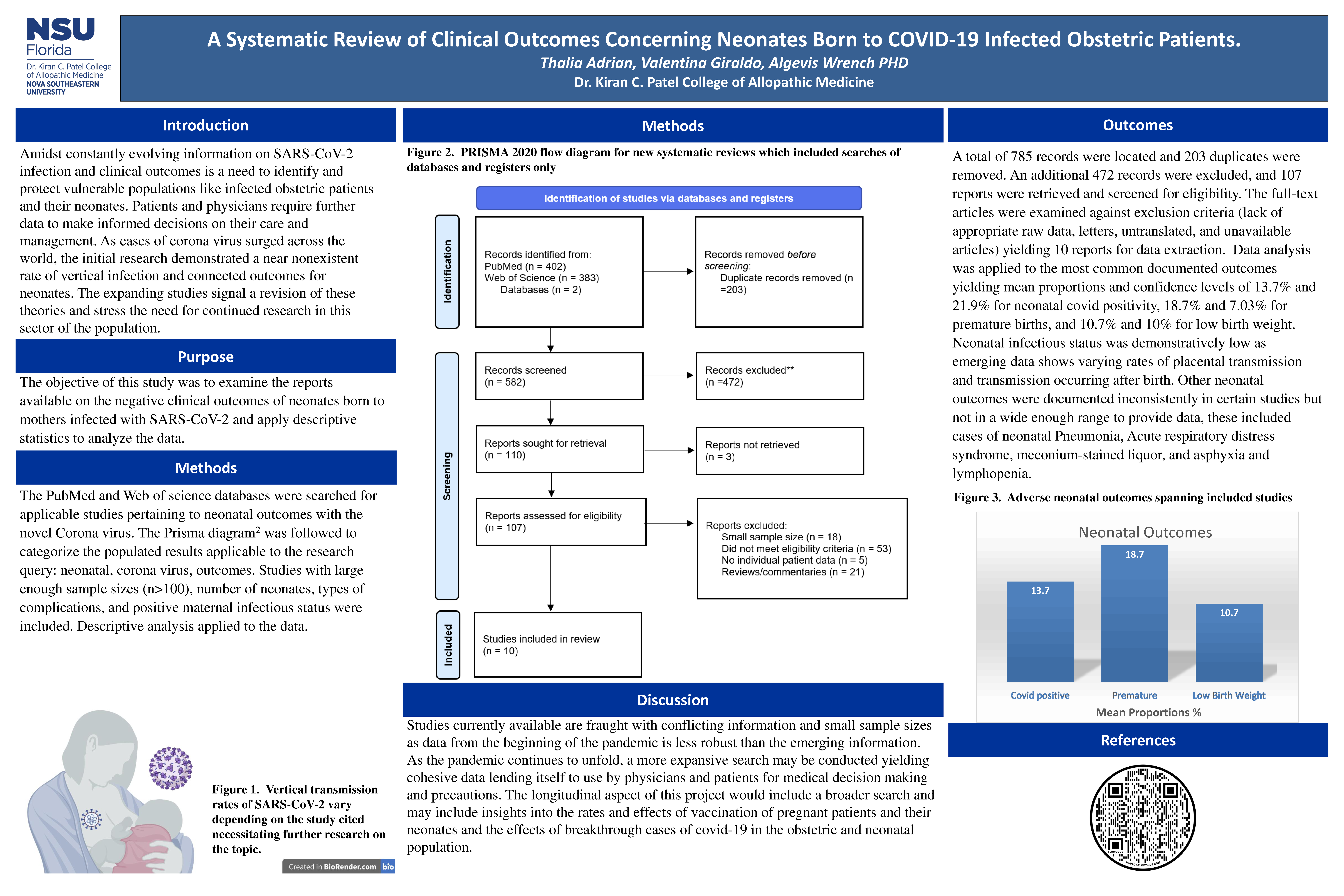
The PubMed and Web of science databases were searched for applicable studies pertaining to neonatal outcomes with SARS-CoV-2. The Prisma diagram1 was applied to the research query: neonatal, corona virus, outcomes. Studies with sample sizes (n>100), number of neonates, types of complications, and positive maternal infectious status were included. Descriptive analysis was applied to the data.
Outcomes
785 (402 PubMed, 383 Web of Science) records were located, and 203 duplicates and an additional 472 records were excluded. The 107 full-text articles were examined against exclusion criteria (lack of data, letters, and unavailable articles) yielding 10 reports for data extraction. Data analysis was applied to the most common documented outcomes yielding mean proportions and confidence levels of 13.7% and 21.9% for neonatal COVID positivity, 18.7% and 7.03% for premature births, and 10.7% and 10% for low birth weight.
Discussion
Studies currently available are fraught with conflicting information and small sample sizes. As the pandemic continues to unfold, a more expansive search may be conducted yielding cohesive data lending itself to use by physicians and patients for medical decision making and precautions.