Abstract
Background
Cancer patients frequently search the Internet for treatment options, and hospital websites are seen as reliable sources of knowledge. Evidence of superiority for proton beam therapy (PBT) exists only in certain disease sites and indications. This study aims to evaluate direct-to-consumer advertising content and claims made by proton therapy centre (PTC) websites worldwide.
Methods
Operational PTC websites in English were identified through the Particle Therapy Co-Operative Group website as of September 1, 2016. Data abstraction of website content was performed independently by two investigators using a standardized form, with discrepancies resolved through consensus. Eight international guidelines were consulted to determine indications for PBT.
Results
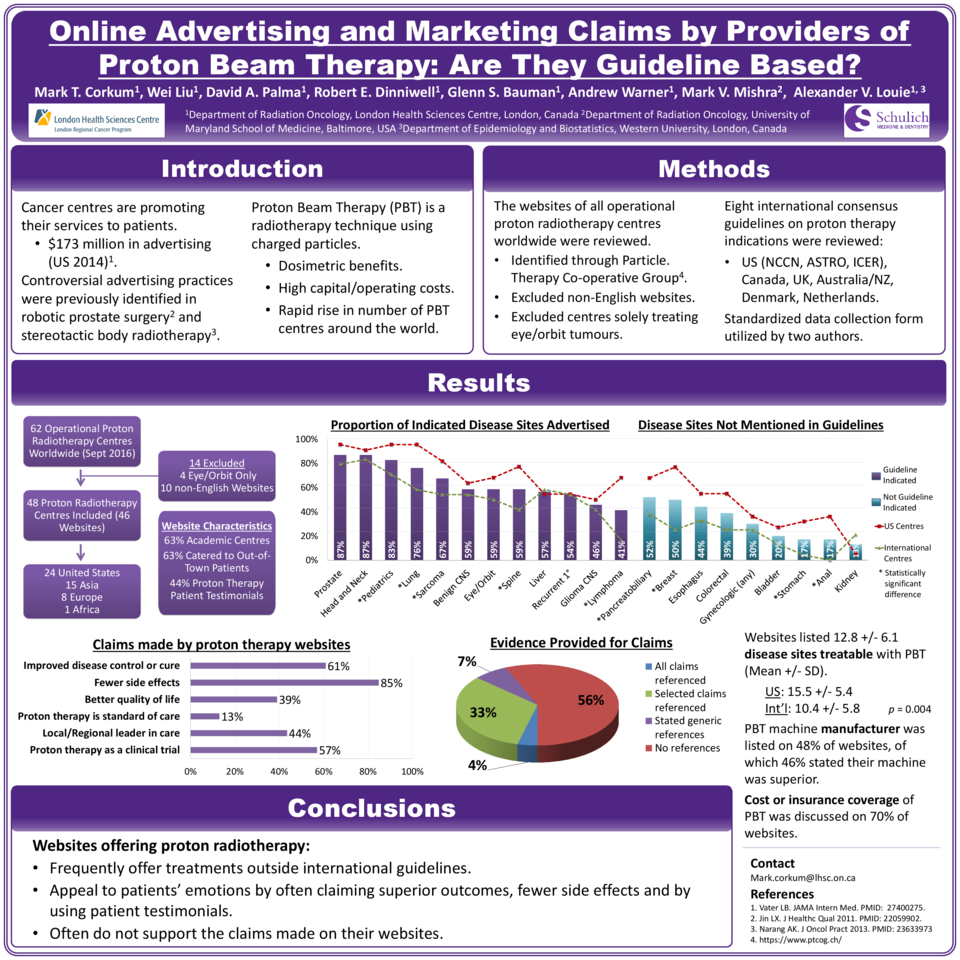
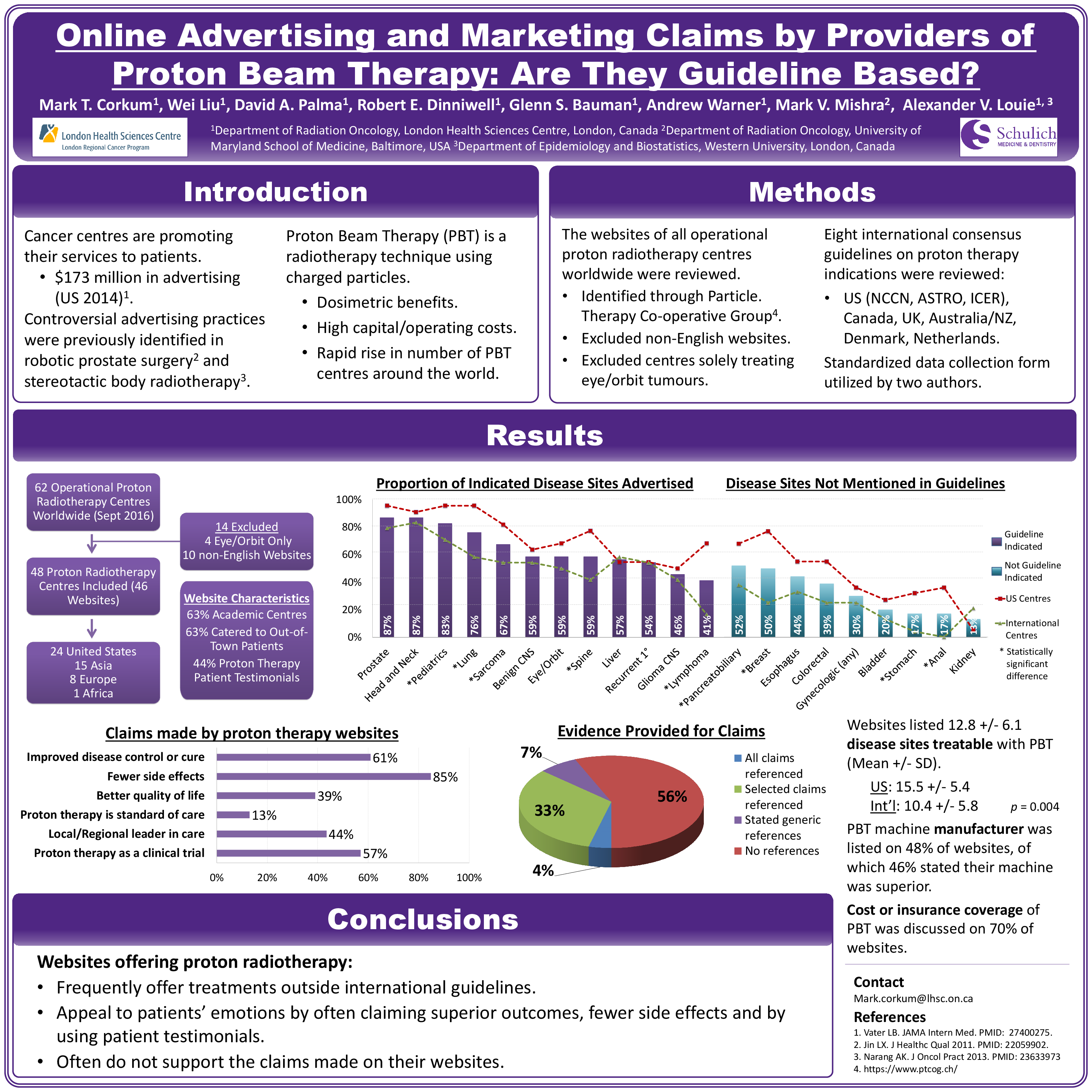
Forty-eight PTCs with 46 English websites were identified. 60·9% of PTC websites claimed proton therapy provided improved disease control or cure. Mean number of treatable disease sites listed were 12·8 ± 6·1 (SD), with US websites listing significantly more indications than international websites (15·5 ± 5·4 vs. 10·4 ± 5·8, p = 0·004). The most common disease sites advertised were prostate (87·0%), head and neck (87·0%) and pediatrics (82·6%), all of which were indications addressed by at least one international guideline. Several disease sites advertised were not present in any consensus guidelines, including pancreatobiliary (52·2%), breast (50·0%), esophagus (43·5%), colorectal (39·1%) and gynecologic (30·4%) cancers.
Conclusion
Information from PTC websites often differs from recommendations found in international consensus guidelines. Inherent challenges exist for emerging medical treatments like PBT in balancing early adoption through increased indications versus generating appropriate evidence prior to widespread implementation. This study highlights the urgent need for comparative effectiveness research to develop high-quality evidence-based recommendations to inform PBT’s judicious use.