Abstract
Purpose:
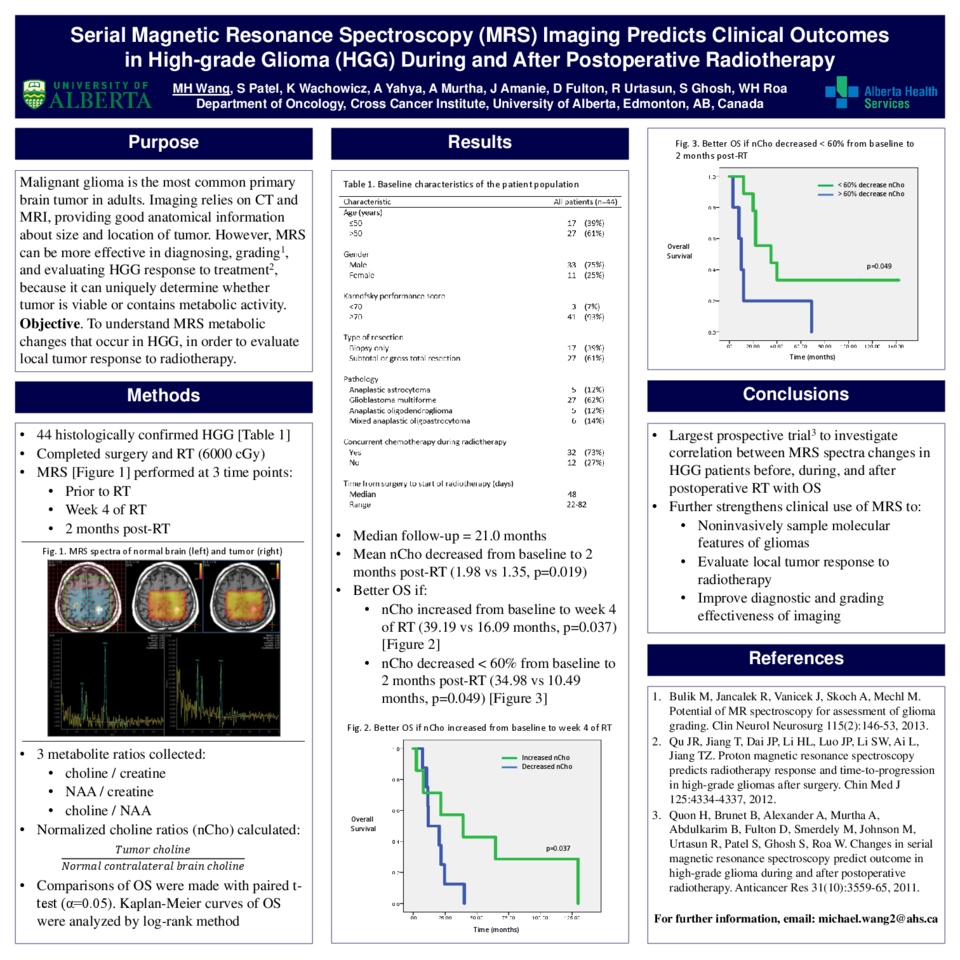
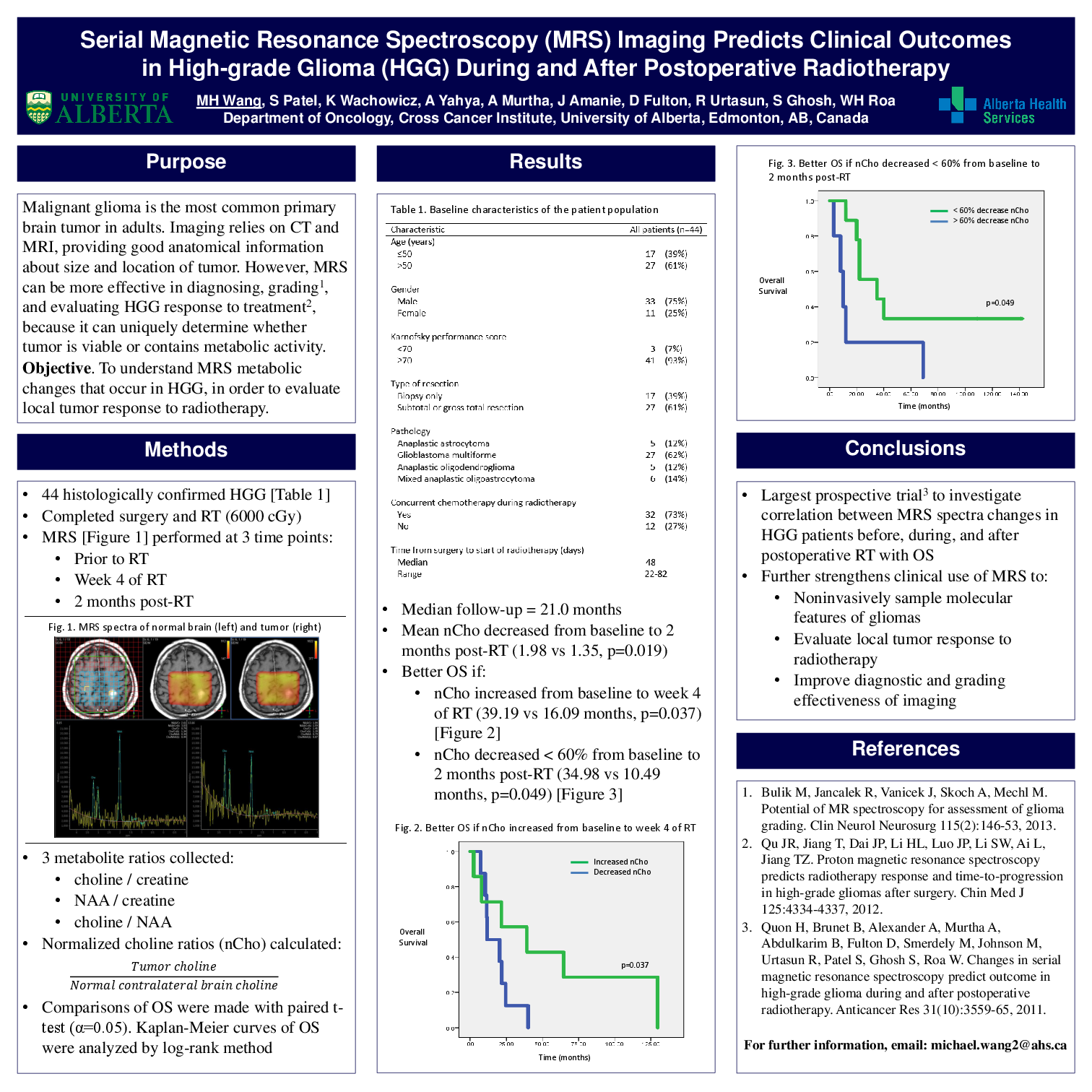
Conventional imaging methods, such as computed tomography (CT) and magnetic resonance imaging (MRI) are not as effective as magnetic resonance spectroscopy (MRS) in diagnosing, grading, and evaluating high-grade glioma (HGG) tumor response to treatment. MRS uniquely determines whether tumor is viable or contains metabolic activity following post-operative radiotherapy (RT). This study’s preliminary data was published, and has currently become the largest prospective trial to investigate the correlation between changes in MRS spectra in HGG patients before, during, and after postoperative RT with overall survival (OS).
Materials and Methods:
Fourty-four patients with histologically confirmed HGG prospectively completed surgery and RT to 6000 cGy. MRS was performed prior to starting RT, during week 4 of RT, and at 2 months post-RT. Metabolite ratios of: 1) choline / creatine; 2) NAA / creatine; and 3) choline / NAA were calculated from MRS spectra at each time point. Normalized metabolite ratios, defined as Tumor metabolite / Normal contralateral brain metabolite, were evaluated for differences in OS from baseline to post-RT. Kaplan-Meier curves of OS were analyzed for statistical significance by the log-rank method, and paired groups of metabolite ratios examining changes over time were compared using the paired t-test.
Results:
After a median follow-up of 21.0 months, mean normalized choline significantly decreased from baseline to 2 months post-RT (1.98 vs 1.35, p=0.019). Patients with <60% decrease in normalized choline from baseline to 2 months post-RT had a significantly better median OS (34.98 vs 10.49 months, p=0.049). Moreover, patients with any increase in normalized choline from baseline to week 4 of RT had significantly better median OS after completing RT (39.19 vs 16.09 months, p=0.037).
Conclusion:
This study is the largest prospective trial to demonstrate that changes in normalized choline from baseline to 2 months post-RT are highly predictive for OS. These prognostic MRS changes further strengthen the clinical use of advanced imaging techniques to improve the diagnostic and grading effectiveness of imaging, to noninvasively sample the molecular features of gliomas, to evaluate local tumor response to radiotherapy, and to optimize individualized treatments for patients with HGG.